Home / Pleth Variability Index (PVi®)
Pleth Variability Index (PVi®)
Pleth Variability Index (PVi®)
An Index for Continuous Noninvasive Dynamic Indication of Fluid Responsiveness in Select Populations of Mechanically Ventilated Adult Patients from a Single Pulse Oximetry Sensor
PVi provides a continuous noninvasive measure of the relative variability in the photoplethysmograph (pleth) during respiratory cycles that may be used as a dynamic indicator of fluid responsiveness in select populations of mechanically ventilated adult patients.1
PVi is available alongside Masimo SET® Pulse Oximetry and rainbow® Pulse CO-Oximetry.
Fluid Optimization
Fluid Optimization
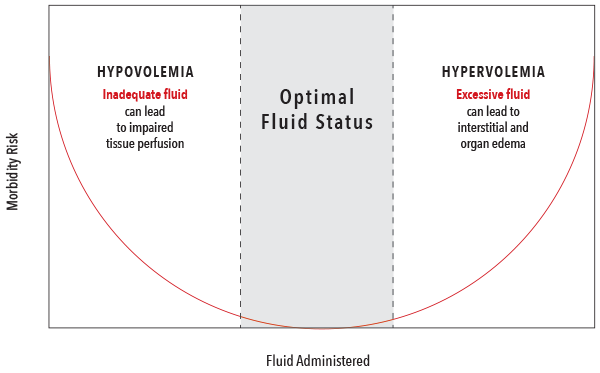
- Fluid administration is one of the most common interventions made to increase cardiac output (CO). However, fluid administration should be balanced to avoid both hypovolemia and hypervolemia, which have both been associated with negative outcomes.2
Identifying Preload Dependence
Identifying Preload Dependence
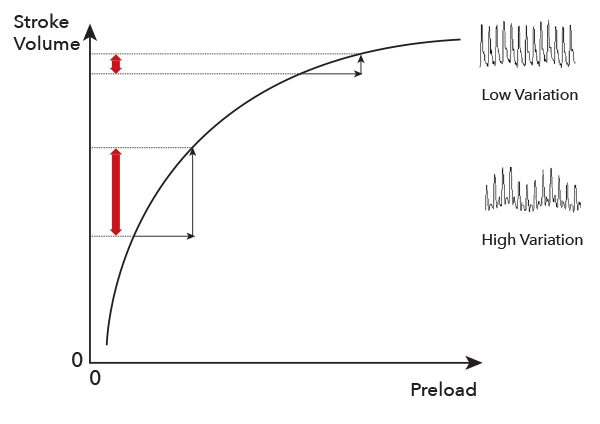
- Preload dependence is defined as the ability of the heart to increase stroke volume in response to an increase in preload/fluid.1
- The Frank-Starling curve defines the relationship between ventricular preload and stroke volume and the preload dependence of fluid responders.1
- During a respiration cycle, changes can be seen in the ventricular filling volume. When the preload/fluid volume is low, these changes in ventricular filling volume result in higher variations in stroke volume.1
Dynamic Parameters
Dynamic Parameters
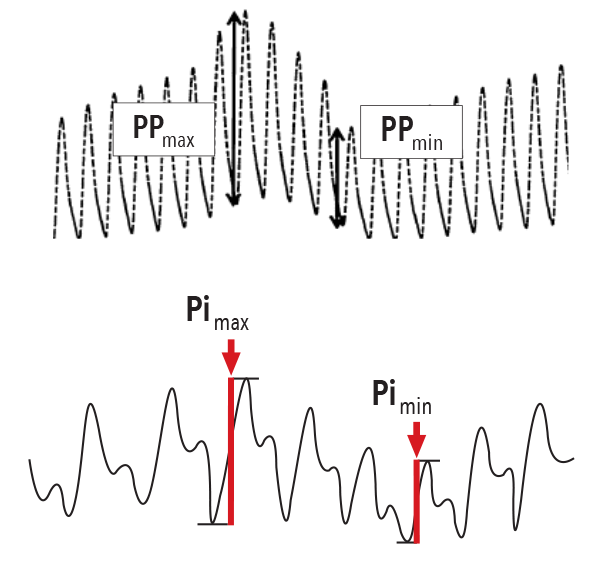
- Multiple hemodynamic parameters (ex. SVV, PPV) have been used to help clinicians assess variations in the arterial pressure waveform resulting from the stroke volume from an indwelling (invasive) arterial cannula or through noninvasive blood pressure cuffs.3
- While dynamic parameters such as PPV and SVV use the arterial pressure waveform, PVi uses the arterial pleth waveform, which reflects the absorption concentration of arterial hemoglobin.3
PVi Technology Overview
PVi Technology Overview
Pleth Variability Index (PVi) is dynamic index between 0-100 which measures the relative variability of the pleth waveform noninvasively detected from a pulse oximetry sensor. It uses the detected pleth amplitudes to automatically calculate the dynamic changes that occur during the respiratory cycle. Higher variability in the pleth waveform has been associated with preload dependence and fluid responders.
This allows PVi to be used as a noninvasive dynamic indicator of fluid responsiveness in select populations of mechanically ventilated adult patients.
The ability of PVi to predict fluid responsiveness can be variable and influenced by numerous patient, procedural, and device-related factors. Fluid management decisions should be based on a complete assessment of the patient’s condition and not solely on PVi.
PVi in Fluid Management Protocols
PVi in Fluid Management Protocols
- Hospital protocols, such as Goal-directed Therapy (GDT) and Enhanced Recovery After Surgery (ERAS), recommend fluid management as part of larger initiatives designed to improve patient care and safety.
- Proper balancing of fluids has been associated with decreased lactate build-up and shorter recovery times.4,5
- Numerous studies have evaluated the utility of PVi as part of various regimens and protocols for fluid management, with varying results and outcomes. For a list of studies, please visit: www.masimo.com/evidence/pulse-oximetry/pvi.
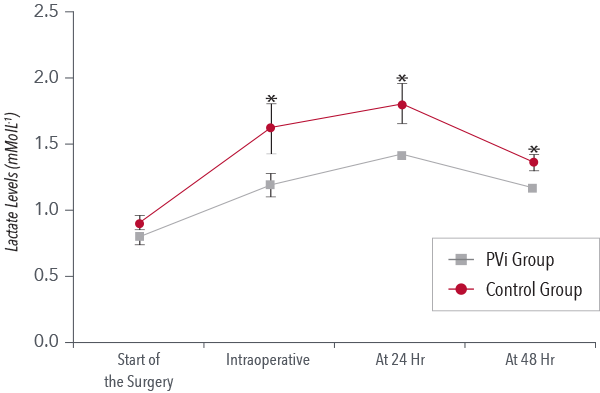
Goal-directed Therapy (GDT)
In a study of eighty-two patients undergoing major abdominal surgery, researchers found that PVi-based goal-directed fluid management reduced the volume of intraoperative fluid infused and reduced intraoperative and postoperative lactate levels.4
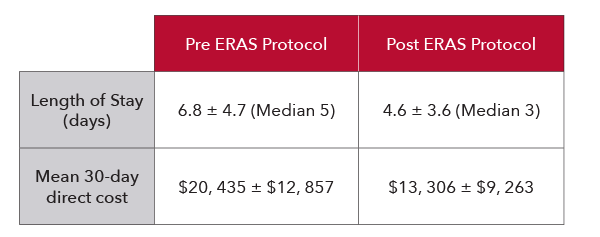
Enhanced Recovery After Surgery (ERAS)
In another study of one hundred and nine patients undergoing colorectal surgery, researchers found that the implementation of an enhanced recovery protocol which included PVi led to improved patient satisfaction and substantial reductions in lengths of stay, complication rates, and costs for patients.5
The GDT and ERAS studies summarized above were conducted on patients undergoing a specific type of procedure and following a specific fluid management protocol. The results may not be reflective of all cases and the described GDT and ERAS protocols may not be appropriate for all types of patients and procedures.
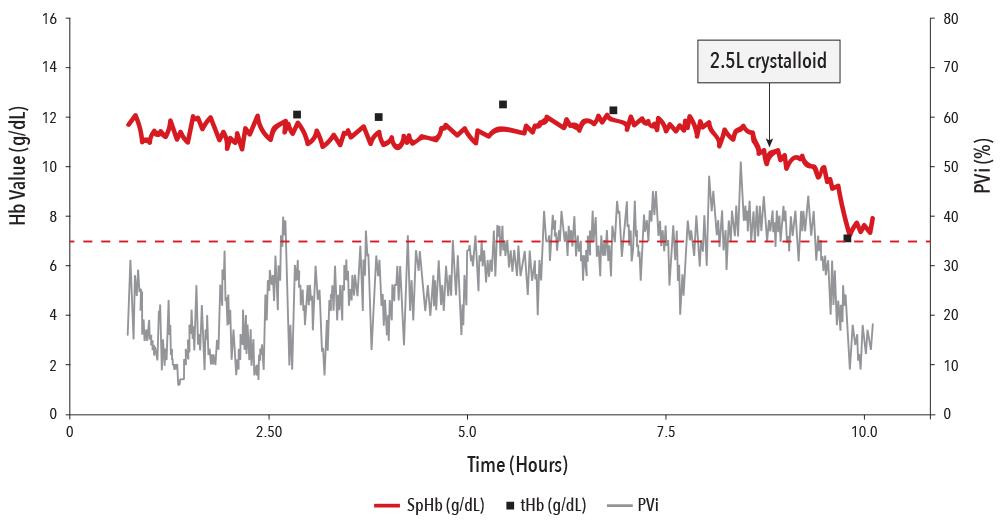
Case Example: PVi Alongside SpHb®
Case Example: PVi Alongside SpHb®
SpHb is a noninvasive continuous parameter for monitoring total hemoglobin and is provided alongside PVi on rainbow® Pulse CO-Oximetry sensors. The following hepatic surgery case depicts the effect of dilution on total hemoglobin upon the infusion of 2.5L of crystalloid.6
PVi and SpHb Support Clinicians in Improving Patient Outcomes
PVi and SpHb Support Clinicians in Improving Patient Outcomes

A single-center quality initiative involving 18,716 patients in the OR, ICU, and PACU was implemented in Limoges, France. The study used a Goal-directed Therapy (GDT) protocol with PVi in conjunction with a blood transfusion protocol based on SpHb. Results demonstrated that monitoring with SpHb and PVi integrated in a vascular filling algorithm was associated with earlier transfusion and reduced 30- and 90-day mortality by 33% and 29%, respectively, on a whole hospital scale.6
References
- 1.
Cannesson et al. Journal of Cardiothoracic Vas Anes. 2010.
- 2.
Bellamy MC. Br J Anaesth. 2006 Dec;97(6):755-7.
- 3.
Chandler et al. J Clin Monit Comput. 2012.
- 4.
Forget P et al. Anesth Analg. 2010 Oct;111(4):910-4.
- 5.
Thiele RH et al. J Am Coll Surg. 2015 Apr;220(4):430-43.
- 6.
Perel Critical Care (2017) 21:291
- 7.
Cros et al. J Clin Monit Comput. Aug 2019: 1-9.
RESOURCES
SpHb is not intended to replace laboratory blood testing. Clinical decisions regarding red blood cell transfusions should be based on the clinician’s judgment considering among other factors: patient condition, continuous SpHb monitoring, and laboratory diagnostic tests using blood samples.
Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. See instructions for use for full prescribing information, including indications, contraindications, warnings, and precautions.





